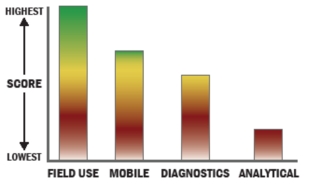
General Description
Envi Assay System is intended for rapid on-site identification of biological material. The system has manual test tickets for Ricin toxin, Botulinum toxin, SEB toxin, Anthrax and Smallpox. The system also contains a reader module that is used to read the test results.
Technical Description
The test tickets are based on Immunoassay technology that uses either gold- or fluorescence labelled antibodies. The reader module has a two-wavelength camera that is capable of reading both labels. The Reader module can be connected to a ChemPro100 Chemical Detector or to a PC.
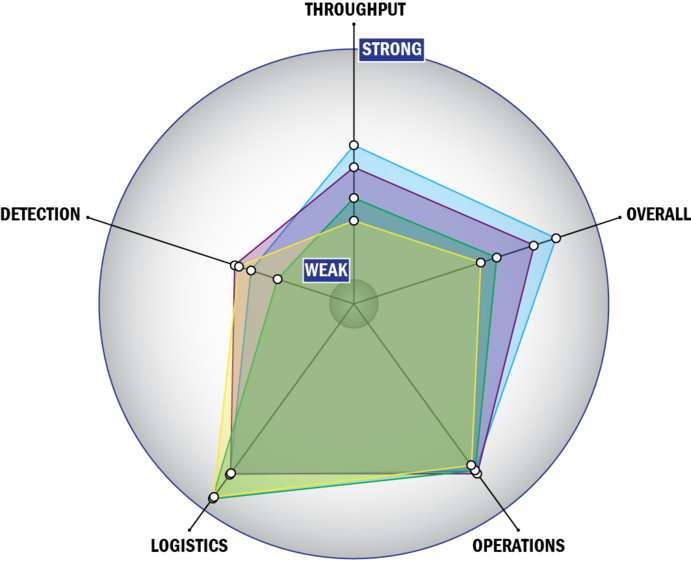
Evaluation Criteria
THROUGHPUT:
- Between 2 and 15 minutes for detection
- 1 sample, single test/sample per run
- 95-32 samples every 2 hours
- The system or approach is not amenable to full or semi-automation
- Device or system is designed for a single use
- 0-1 solutions, buffer, eluents, and/or reagents
- 3 components
- Less than 5 minutes is required for set-up
- 3-5 steps are required for detection
LOGISTICS:
- Very brief (minutes-hours) training and minimal technical skills
- Approximately the size of a soda can
- Less than 1 kg
- System or device uses batteries
- 4-8 hours battery life
OPERATIONS:
- Can be used from 4°C to 41°C
- Components must be stored at room temperature (27°C)
- Performance is not influenced by relative humidity
- Between 1 to 3 years shelf life
- Results cannot be viewed in real-time
- The system is not capable of autonomy
- The system software is open and available for modification
- The system hardware is open and available for modification
DETECTION:
- Not possible for the system to achieve 510K clearance
- Not possible for the system to achieve FDA approval
- Less than 10 µL
- Good specificity. System has a consistently low level of false alarms (2-5%)
- Greater than 100,000 CFU per mL
- Greater than 100,000 PFU per mL
- 10-100 ng per mL
- Spore lysis not necessary for detection by system




 Top Tier
Top Tier Second Tier
Second Tier Third Tier
Third Tier Fourth Tier
Fourth Tier Bottom Tier
Bottom Tier