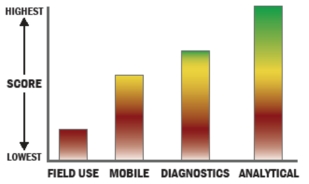
General Description
The Food-Safety Security Module (FSSM) is a scalable, integrated package that utilizes a commercial mass spectrometer, commercial AP-MALDI, and commercial databases to detect and identify food-borne pathogens in less than 24-hours total time; including enrichment. Processing can be either manual or automated with use of the supplemental auto-processing module. In terms of logistics, the consumables cost per sample is less than one dollar and train-up for non-technical users is one day.
Technical Description
The Food-Safety Security Module (FSSM) builds on our Wide-Spectrum Bio-ID technologies and uses a commercial ion-trap mass spectrometer in which MS/MS is conducted via AP-MALDI of biological samples after enrichment. Biological materials undergo single-spot proteomic processing and MS/MS targeted bio-marker analysis to sift rapidly thru clutter against a commercial internet-accessible database such as MASCOT.
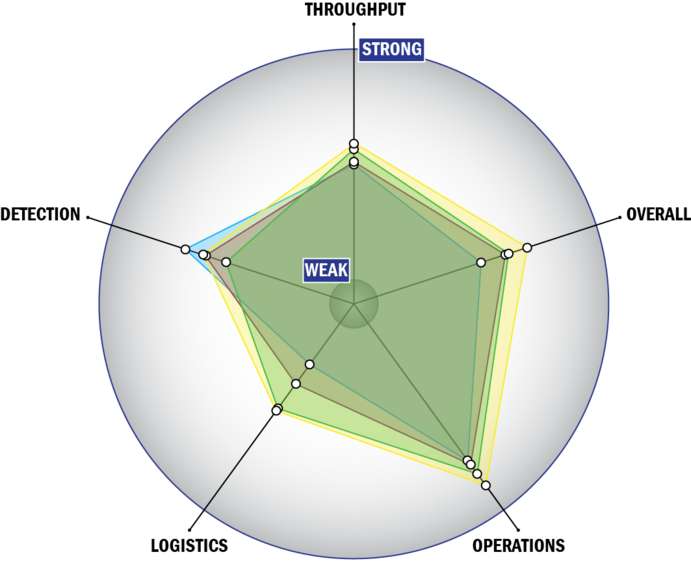
Evaluation Criteria
THROUGHPUT:
- Between 2 and 15 minutes for detection
- Multiple samples, multiple tests/sample per run
- 95-32 samples every 2 hours
- The system or device is currently fully automated
- Device or system is intended for multiple detection assays
- 3 solutions, buffer, eluents, and/or reagents
- 1 component
- 10-20 minutes is required for set-up
- 3-5 steps are required for detection
LOGISTICS:
- An afternoon of training and some technical skills required
- Approximately the size of a home dishwasher
- More than 50 kg
- Wireless and wired connections are available
- System or device has 110V electrical requirement
OPERATIONS:
- Can be used from 25°C to 37°C
- Components must be stored at 4°C
- Performance is not influenced by relative humidity
- Between 1 to 3 years shelf life
- Greater than 10 years expected life
- Results can be viewed in real-time
- The system is not capable of autonomy
- The system software is open and available for modification
- The system hardware is open and available for modification
DETECTION:
- Possible the system could receive 510K clearance, no current efforts at this time
- Possible the system could receive FDA approval, no current efforts at this time
- Less than 10 µL
- Excellent specificity. System has occasional false alarms under certain conditions (<2%)
- 1,000-10,000 CFU per mL
- 1,000-10,000 PFU per mL
- 10-100 ng per mL
- Fully automated spore lysis




 Top Tier
Top Tier Second Tier
Second Tier Third Tier
Third Tier Fourth Tier
Fourth Tier Bottom Tier
Bottom Tier