General Description
Advanced Liquid Logic, Inc (ALL) is committed to the development of cost-effective automation solutions for complex bioassay workflows. Our proprietary digital microfluidics technology is broadening the availability of molecular diagnostics, enabling new-to-the-world bioanalytical products and accelerating & improving life science research. ALL has developed a patented liquid handling technology called “digital microfluidics’ that drastically reduces the cost, complexity and time required for fully automated sample-in/answer-out workflows. The key factors that differentiate the ALL system are: - Assay flexibility – adding and optimizing assay types is rapidly accomplished by editing software
- Low cost – component manufacturing processes are cost-effective and well-established
- Broadly deployable – suited to any setting, from research lab to zero-resource field environment
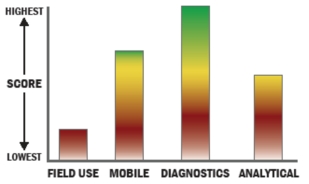
An exhaustive range of assay formats, relevant to clinical diagnostics and general life sciences research, have been translated to ALL’s digital microfluidics technology. Some of the assays that have been implemented on our system include: - Sample-in/answer-out (qPCR, immunoassays & DNA sequencing)
- Sample extraction & purification (from crude samples to pure: DNA, RNA or protein)
- Next-gen sequencing template prep
- Real-time and Endpoint qPCR
- Immunoassays
- DNA sequencing (pyrosequencing)
- Newborn screening (enzymatic activity assays)
- Cell-based assays
- Clinical chemistry
- Coagulation monitoring
Technical Description
The instrument and cartridge-based platform developed by ALL eliminates the need for cumbersome pumps, valves and tubes by manipulating liquid droplets on an array of electrodes. The so-called “electrowetting” effect provides a motive force that is flexibly controlled through software. In conventional microfluidics liquid handling is directed by networks of tubes or channels that must be redesigned for every unique application. ALL's digital microfluidic systems shatter this traditional one device-one workflow paradigm. Within the ALL cartridge, electrode arrays are arranged on inexpensive printed circuit boards (PCBs). An injection molded plastic plate above the PCB creates a chamber where droplets are manipulated. Reagents are pre-placed in the cartridge at the time of manufacture to enable minimally-trained personnel to run complex bioanalytical protocols.
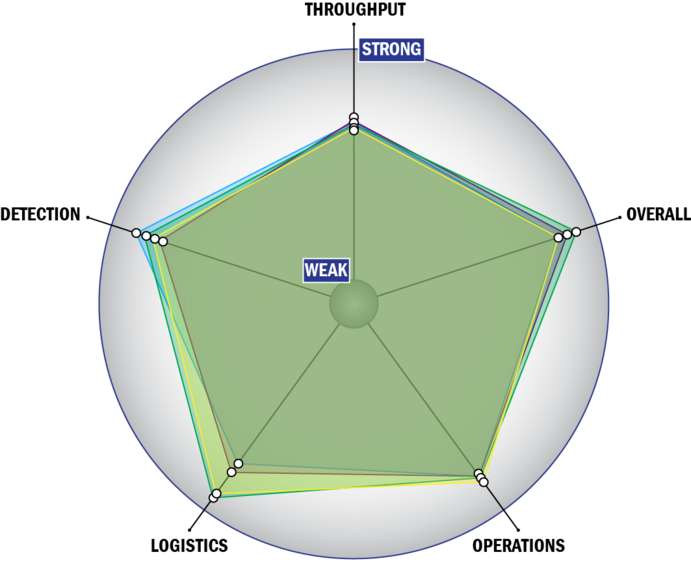
Evaluation Criteria
THROUGHPUT:
- Between 15 and 30 minutes for detection
- Multiple samples, multiple tests/sample per run
- 95-32 samples every 2 hours
- The system or device is currently fully automated
- Device or system is intended for multiple detection assays
- 3 solutions, buffer, eluents, and/or reagents
- 1 component
- Less than 5 minutes is required for setup
- 1-2 steps are required for detection
LOGISTICS:
- Very brief (minutes-hours) training and minimal technical skills
- Approximately the size of a toaster
- Between 5 and 25 kg
- Wired connections are available
- System or device uses batteries
- 4-8 hours battery life
OPERATIONS:
- Can be used from 4°C to 41°C
- Components must be stored at room temperature (27°C)
- Performance is not influenced by relative humidity
- Between 6 months and 1 year shelf life
- 3-5 years expected life
- Results can be viewed in real-time
- The system could easily be adapted into a fully autonomous
- The system software is open but modification requires licensing
- The system hardware is open but modification requires licensing
DETECTION:
- Efforts are underway to achieve 510K clearance
- Efforts are underway to achieve FDA approval
- Less than 10 µL
- Excellent specificity. System has occasional false alarms under certain conditions (<2%)
- 1-100 CFU/mL
- 100-1,000 PFU/mL
- Less than 1 ng/mL
- Add on capability that is full or semi-automated for spore lysis




 Top Tier
Top Tier Second Tier
Second Tier Third Tier
Third Tier Fourth Tier
Fourth Tier Bottom Tier
Bottom Tier