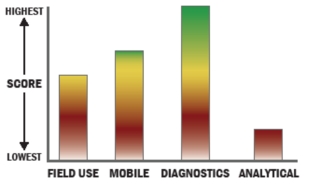
General Description
The iScan System is a cutting-edge, dedicated array scanner that supports rapid, sensitive, and accurate imaging of Illumina's array-based genetic analysis products. The system features a high signal-to-noise ratio, high sensitivity, an excellent limit of detection, and a broad dynamic range leading to exceptionally high data quality. The iScan System supports Infinium, GoldenGate, DASL, Gene Expression (Direct Hybridization), and Methylation assays along with different multi-sample BeadChip formats.
Technical Description
At the System's core is the iScan Reader, which incorporates high-performance lasers, optics, and detection systems. The iScan Reader offers sub-micron resolution and unmatched throughput rates. Even the highest density BeadChips can be scanned in just minutes, allowing processing of up to 96 multi-sample BeadChips per day. These imaging features are especially beneficial for high-density genotyping, CNV analysis, DNA methylation, and gene expression profiling.
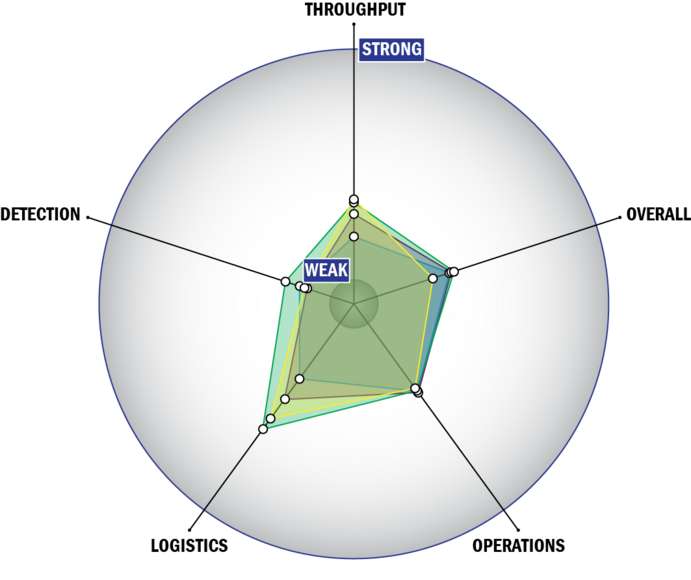
Evaluation Criteria
THROUGHPUT:
- Greater than 8 hours for detection
- Multiple samples, multiple tests/sample per run
- Less than 32 samples every 2 hours
- The system or device is currently semi-automated
- Device or system is intended for multiple detection assays
- 5 or more solutions, buffer, eluents, and/or reagents
- 5 or more components
- Less than 5 minutes is required for set-up
- Greater than 12 steps are required for detection
LOGISTICS:
- A day of training and technical skills are required
- Approximately the size of a home dishwasher
- More than 50 kg
- System or device has 110V electrical requirement
OPERATIONS:
- Can be used from 25°C to 37°C
- Components must be frozen (-20°C)
- Performance is not influenced by relative humidity
- Between 1 to 6 months shelf life
- Greater than 10 years expected life
- Results cannot be viewed in real-time
- The system could be adapted to a fully autonomous system with some effort
- The system software is closed and not available for modification
- The system hardware is closed and not available for modification
DETECTION:
- Possible the system could receive 510K clearance, no current efforts at this time
- Possible the system could receive FDA approval, no current efforts at this time
- This system does not test liquids
- Excellent specificity. System has occasional false alarms under certain conditions (<2%)
- Manual kit not integrated with the system handles spore lysis




 Top Tier
Top Tier Second Tier
Second Tier Third Tier
Third Tier Fourth Tier
Fourth Tier Bottom Tier
Bottom Tier