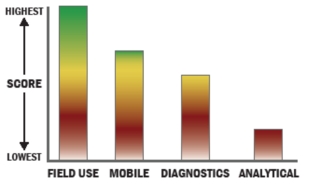
General Description
A protocol has been optimized to enable rapid enrichment, concentration, purification and polymerase chain reaction (PCR) detection of pathogenic organisms. While the developed protocol is specifically for Listeria monocytogenes, other pathogenic organisms could also be processed with variations to the protocol. The sample preparation and detection approach is amenable to multiplexing to allow detection of multiple pathogens (bacterial and viral) in a single sample. The protocol was developed specifically for analyzing surface swabs from food processing plant surfaces, but could be adapted to other surfaces, bodily fluids, environmental samples, and food.
Technical Description
We have investigated and developed unique enrichment broths to enable rapid enrichment (4-6 hours) of pathogenic organisms. We have also developed protocols to remove debris and interfering substances using a combination of filtration and immunomagnetic beads, which also provide a high degree of pathogen concentration and sample purification. We have also optimized PCR detection of Listeria monocytogenes.
Evaluation Criteria
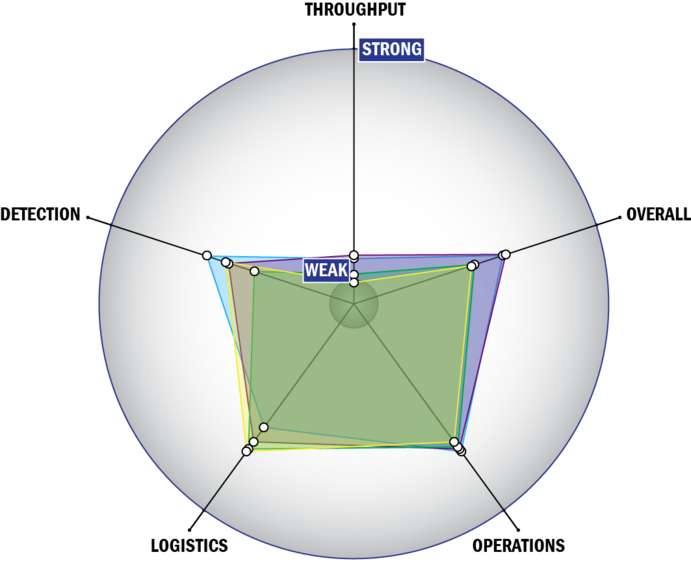
THROUGHPUT:
- Between 60 minutes and 8 hours for detection
- 1 sample, single test/sample per run
- Less than 32 samples every 2 hours
- The system could be adapted to a semi-automated system with some effort
- Device or system is intended for multiple detection assays
- 4 solutions, buffer, eluents, and/or reagents
- 5 or more components
- Less than 5 minutes is required for set-up
- Greater than 12 steps are required for detection
LOGISTICS:
- An afternoon of training and some technical skills required
- Approximately the size of a carry-on luggage suitcase
- Between 5 and 25 kg
- Satellite, wireless and wired connections are available
- System or device has 110V electrical requirement
OPERATIONS:
- Can be used from 4°C to 41°C
- Components must be stored at room temperature (27°C)
- Performance is not influenced by relative humidity
- Between 6 months and 1 year shelf life
- 3-5 years expected life
- Results can be viewed in real-time
- The system could be adapted to a fully autonomous system with significant effort
- The system software is open and available for modification
- The system hardware is open and available for modification
DETECTION:
- Possible the system could receive 510K clearance, no current efforts at this time
- Possible the system could receive FDA approval, no current efforts at this time
- Less than 10 µL
- Excellent specificity. System has occasional false alarms under certain conditions (<2%)
- 1-100 CFU per mL
- 1-100 PFU per mL
- Manual kit not integrated with the system handles spore lysis




 Top Tier
Top Tier Second Tier
Second Tier Third Tier
Third Tier Fourth Tier
Fourth Tier Bottom Tier
Bottom Tier