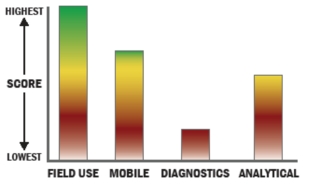
General Description
Economical, rapid (i.e., near real time), sensitive, and specific detection or diagnosis of exposure to biowarfare agents (i.e., bacteria, virus, toxins). With little or no sample-prep and minimal training required, the shirt-pocket size "reader" with disposable sample cartridge facilitates use in the field (including austere environments) or point-of-care settings. Supports single analyte or multiplex testing, in complex matrices including environmental or human derived samples.
Technical Description
Our device is based on surface acoustic wave (SAW)-based sensor technology that directly detects virus, bacteria, or toxin through binding of the target agent to an antibody or peptide coupled to a piezoelectric surface. This binding results in a change to the electrical transfer function of the device which can be measured at its output. Sensitivity and selectivity are conferred through the use of selected ligands.
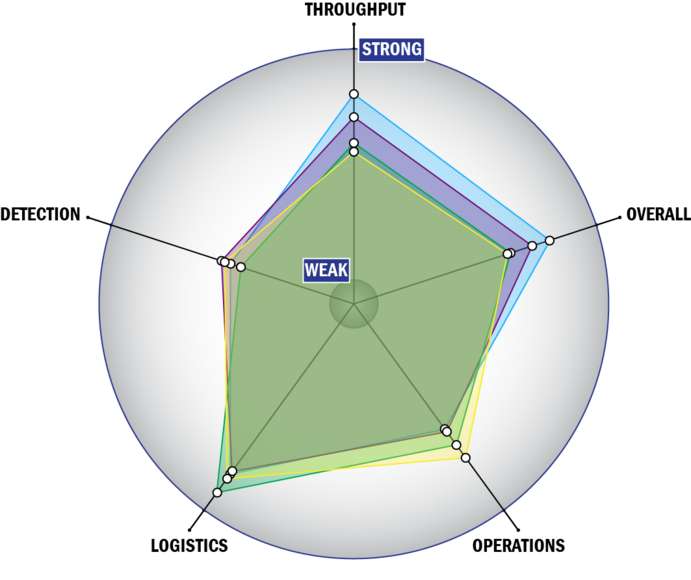
Evaluation Criteria
THROUGHPUT:
- 2 minutes or less for detection
- 1 sample, <10 tests/sample per run
- Less than 32 samples every 2 hours
- The system or device is currently semi-automated
- Device or system is intended for multiple detection assays
- 0-1 solutions, buffer, eluents, and/or reagents
- 1 component
- Less than 5 minutes is required for set-up
- 1-2 steps are required for detection
LOGISTICS:
- Very brief (minutes-hours) training and minimal technical skills
- Approximately the size of a toaster
- Between 1 and 5 kg
- Wireless and wired connections are available
- System or device uses batteries
- 4-8 hours battery life
OPERATIONS:
- Can be used from 4°C to 41°C
- Components must be stored at room temperature (27°C)
- Between 1 to 6 months shelf life
- Greater than 10 years expected life
- Results can be viewed in real-time
- The system could be adapted to a fully autonomous system with significant effort
- The system software is closed and not available for modification
- The system hardware is closed and not available for modification
DETECTION:
- Possible the system could receive 510K clearance, no current efforts at this time
- Possible the system could receive FDA approval, no current efforts at this time
- Less than 250 µL
- 1-100 CFU per mL
- 100-1,000 PFU per mL
- Spore lysis not necessary for detection by system




 Top Tier
Top Tier Second Tier
Second Tier Third Tier
Third Tier Fourth Tier
Fourth Tier Bottom Tier
Bottom Tier