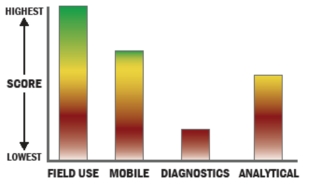
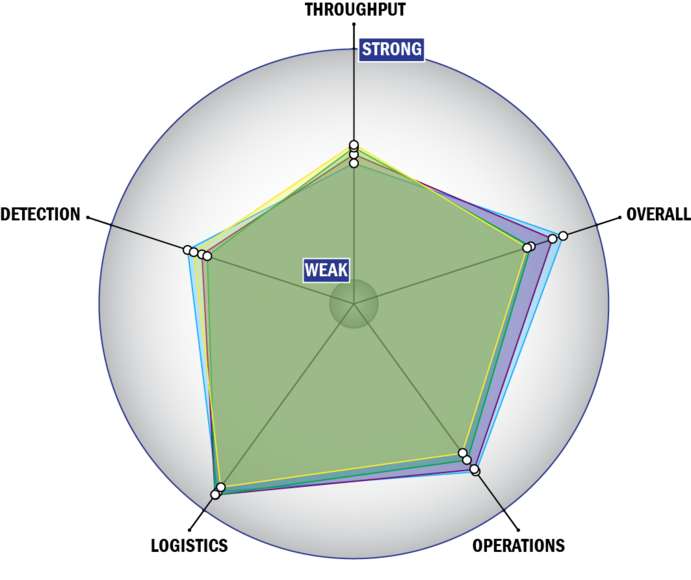
General Description
T2’s revolutionary new technology is based on clinically-proven magnetic resonance MR technology, and uses nanoparticles coupled with reagents to quickly detect—within minutes—the presence of specific substances in solution using a miniaturized, portable MRI instrument. Detection of the high level magnetic resonance signal from the solution enables the detection of low concentrations of target agents or substances. Unlike most existing diagnostic detection techniques which are based on optical detection methods that require pure samples and multiple processing steps, T2’s technology is not optical and therefore does not require purification of biological samples. This significant advantage allows the T2 system to perform single-step processing and rapid turnaround times without the need for trained technicians. Furthermore, the technology can accurately identify almost any specimen, including proteins, nucleic acids, or enzymes; bacterial, cancer or other cells; viruses; or small molecule drug compounds within almost any sample, including whole blood, plasma, serum and urine. T2’s system is faster, more reliable, more portable and more readily accessible than any diagnostic testing system now on the market.
Technical Description
Compact, rugged, non-optical detection of immunoassay and nucleic acid assay targets, directly in unprocessed samples.
Evaluation Criteria
THROUGHPUT:
- Between 2 and 15 minutes for detection
- Multiple samples, multiple tests/sample per run
- The system or device is currently fully automated
- Device or system is intended for multiple detection assays
- 4 solutions, buffer, eluents, and/or reagents
- 1 component
- Greater than 20 minutes is required for set-up
- 1-2 steps are required for detection
LOGISTICS:
- Very brief (minutes-hours) training and minimal technical skills
- Approximately the size of a toaster
- Between 1 and 5 kg
- Satellite, wireless and wired connections are available
- System or device has 110V electrical requirement
- 4-8 hours battery life
OPERATIONS:
- Can be used from < -21°C to > 42°C (All temperatures)
- Components must be stored at room temperature (27°C)
- Performance is not influenced by relative humidity
- Between 1 to 3 years shelf life
- 1-3 years expected life
- Results cannot be viewed in real-time
- The system could be adapted to a fully autonomous system with some effort
- The system software is open but modification requires licensing
- The system hardware is open but modification requires licensing
DETECTION:
- Possible the system could receive 510K clearance, no current efforts at this time
- Possible the system could receive FDA approval, no current efforts at this time
- Less than 10 µL
- 1-100 CFU per mL
- 1-100 PFU per mL
- Less than 1 ng per mL
- Fully automated spore lysis
- < 1x10-6 mg/m3
- < 1 ppb




 Top Tier
Top Tier Second Tier
Second Tier Third Tier
Third Tier Fourth Tier
Fourth Tier Bottom Tier
Bottom Tier