General Description
The PanNAT system is a sample-to-solution, man-portable, PCR-based instrument that is mains and/or battery powered, lightweight and WiFi-enabled. It processes a microfluidics-enabled cartridge into which all reagents are integrated for extraction, amplification and detection of multiple targets from a single sample. Waste is captured on cartridge. Designed for lowest cost, the system employs molecular beacon probe technology and BTI dyes and quenchers together with Micronics’ novel heating and cooling method for end point detection. It is real time capable. The system is designed for use at ambient temp; no refrigeration is required. The system is designed to meet the FDA’s 510(k) Class II device and CLIA Waiver guidelines.
Technical Description
Microfluidics-enabled sample processing of all nucleic acid assay steps with reagents integrated into the disposable cartridge for multiplex end point polymerase chain reaction detection in a man-portable point of care instrument. Novel heating and cooling platform and lowest cost fluorescent detection.
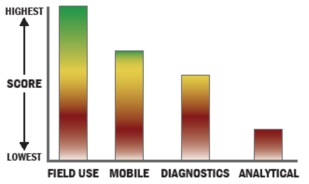
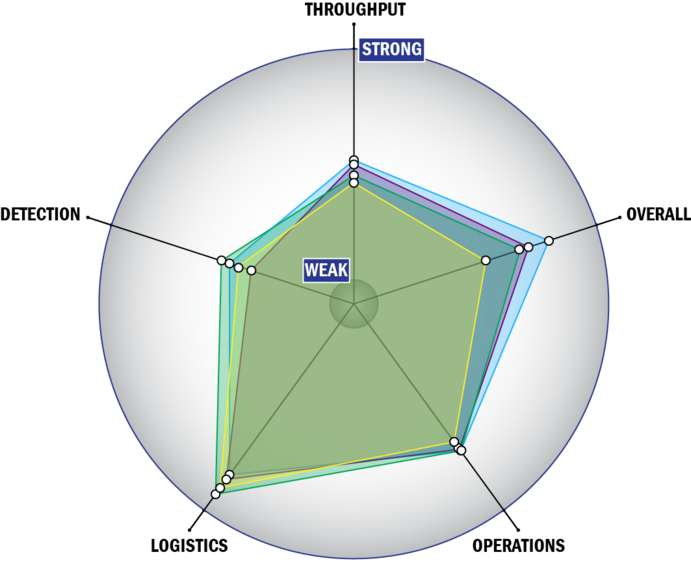
Evaluation Criteria
THROUGHPUT:
- Between 30 and 60 minutes for detection
- 1 sample, >10 tests/sample per run
- Less than 32 samples every 2 hours
- The system or device is currently fully automated
- Device or system is intended for multiple detection assays
- 4 solutions, buffer, eluents, and/or reagents
- 1 component
- Less than 5 minutes is required for setup
- 1-2 steps are required for detection
LOGISTICS:
- Very brief (minutes-hours) training and minimal technical skills
- Approximately the size of a toaster
- Between 1 and 5 kg
- Wireless and wired connections are available
- System or device uses batteries
- 4-8 hours battery life
OPERATIONS:
- Can be used from 4°C to 41°C
- Components must be stored at room temperature (27°C)
- Performance is not influenced by relative humidity
- Between 1 to 3 years shelf life
- Results cannot be viewed in real-time
- The system is not capable of autonomy
- The system software is open but modification requires licensing
- The system hardware is closed and not available for modification
DETECTION:
- Possible the system could receive 510K clearance, no current efforts at this time
- Possible the system could receive FDA approval, no current efforts at this time
- Less than 250 µL
- Excellent specificity. System has occasional false alarms under certain conditions (<2%)
- 100-1,000 CFU per mL
- 100-1,000 PFU per mL




 Top Tier
Top Tier Second Tier
Second Tier Third Tier
Third Tier Fourth Tier
Fourth Tier Bottom Tier
Bottom Tier