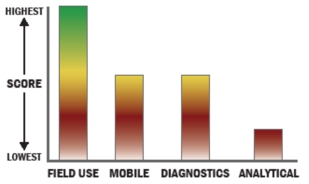
General Description
Biomeme's platform turns any smartphone or like device (iPod Touch, iPad Mini, etc.) into a mobile lab for performing advanced diagnostics. The system requires no special lab equipment or experience and can be used at the point of need in the field or in a mobile lab. The full system includes a hardware add-on, a mobile device software application, and disposable test kits. The system performs molecular diagnostics and near real-time surveillance of pathogens via real-time quantitative polymerase chain reaction (qPCR). In addition to Biomeme's tests, the platform is open enough for experienced users in analytical and diagnostic laboratories to develop their own tests for use on the system.
Technical Description
Real time hydrolysis probe polymerase chain reaction amplification of genetic material on a smartphone or like device (iPod Touch, iPad Mini) hardware add-on thermal cycler.
Notes:
Being tested as part of the DoD's JUPITR ATD.
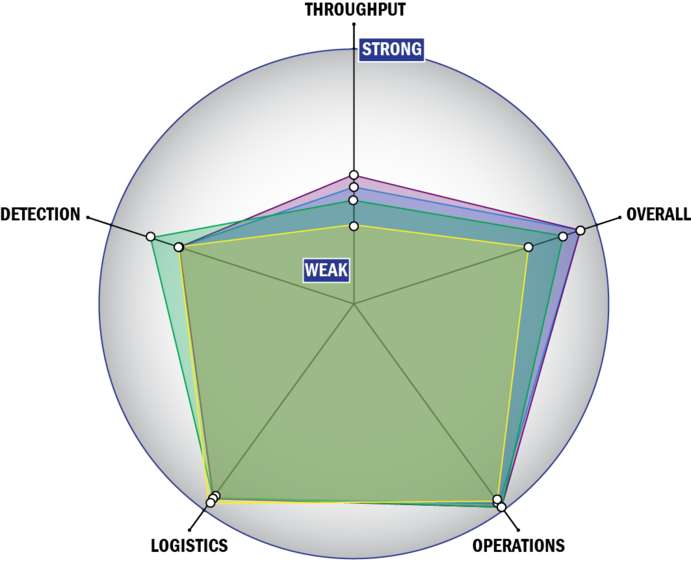
Evaluation Criteria
THROUGHPUT:
- Between 30 and 60 minutes for detection
- 1 sample, <10 tests/sample per run
- Less than 32 samples every 2 hours
- The system could be adapted to a fully automated system with some effort
- Device or system is intended for multiple detection assays
- 4 solutions, buffer, eluents, and/or reagents
- 4 components
- No set-up of the system is required
- 3-5 steps are required for detection
LOGISTICS:
- Very brief (minutes-hours) training and minimal technical skills
- Approximately the size of a soda can
- Less than 1 kg
- Satellite, wireless and wired connections are available
- System or device has 110V electrical requirement
- 4-8 hours battery life
OPERATIONS:
- Components must be stored at room temperature (27°C)
- Performance is not influenced by relative humidity
- Between 1 to 3 years shelf life
- Greater than 10 years expected life
- Results can be viewed in real-time
- The system could be adapted to a fully autonomous system with some effort
- The system software is open but modification requires licensing
- The system hardware is open but modification requires licensing
DETECTION:
- Efforts are underway to achieve 510K clearance
- Efforts are underway to achieve FDA approval
- Less than 100 µL
- Excellent specificity. System has occasional false alarms under certain conditions (<2%)
- 1-100 CFU per mL
- 1-100 PFU per mL
- Manual kit not integrated with the system handles spore lysis




 Top Tier
Top Tier Second Tier
Second Tier Third Tier
Third Tier Fourth Tier
Fourth Tier Bottom Tier
Bottom Tier